|
4/10/2024 0 Comments Water density in gml
(Wikipedia) Converter You are currently converting density units from gram per millilitre to pound per gallon (U.S.) 1 g/mL 8.3454063545262 lb/gal. Specific gravity has no units, because the density units cancel in its calculation.\)Īfter applying the correct number of significant figures, the density of acetic acid would be reported as 1.049 g/mL. The density of a material is defined as its mass per unit volume. Generally, water is the reference substance for comparing solids and liquids, and air is the reference substance for comparing gases.Ī value of specific gravity must state the temperature at which the densities were measured. The specific gravity (sp gr) of a substance is the ratio of its density to that of a reference substance: Often, particularly in discussing fluids, specific gravity is reported rather than density. 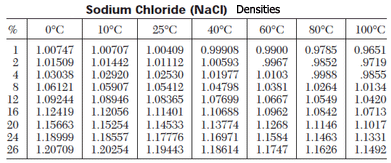
The three quantities (mass, volume, and density), you can calculate the third. TABLE 2.8 Densities of some common solids and liquids underĭensity is a conversion factor that relates mass to volume. Mercury is one of the heaviest liquids known. The density of glycerine is an important physical property that helps determine its suitability for different applications. Except for water, the densities in Table 2.8 are given at 0☌.įIGURE 2.6 The density of mercury (13.6 g/mL) compared with the density of water (1.000 g/mL). Glycerine density refers to the measurement of the mass per unit volume of glycerine, a colorless and odorless liquid commonly used in various industries. For example, the density of water at 4☌ is 1.000 g/mL and at 80☌ is 0.9718 g/mL the density of oxygen is 1.43 g/L at 0☌ and 1.10 g/L at 80☌. For example, heres how to convert 5 grams to milliliters for an ingredient or substance with a density of 0.7 g/mL. For example, the density of gasoline is about 30% less than that of water, and the density of chloroform is about 50% greater.ĭensities vary with temperature. A common unit of measurement for waters density is gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3). Thus, the volume in milliliters is equal to the weight in grams divided by the density (in g/mL) of the ingredient, substance, or material. The densities of liquids vary some are less dense than water, whereas others are more dense. The densities of most metals are greater than that of water. Based on the information in Table 2.8, we can make some basic observations. Using these units, the density of water is given as 1.000 g/mL at 4☌. Recall from Table 2.1 that 1 mL = 1 cm 3. The densities of solids are usually given in grams per cubic centimeter (g/cm 3), the densities of gases in grams per liter (g/L), and the densities of liquids in grams per milliliter (g/mL). 
Table 2.8 lists the densities of some common solids and liquids under normal conditions. Density is a physical property that characterizes and identifies a particular kind of matter (see Figure 2.6). TABLE 2.7 Mass, volume, and density of iron samplesĪll samples of the same kind of matter under the same conditions have the same density. 
However, neither their masses nor their volumes show that all the samples are iron, but all the samples do have the same ratio of mass to volume, as is shown in the far right column. We can measure the mass and volume of a sample, as was done for several samples of iron with results shown in Table 2.7. We have said that chemists determine the properties of matter, particularly those properties that help identify the composition of a sample. Hematite nanostructures are strong candidates for the development of sustainable water splitting technologies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
